Download the PDF Version of this article.
Cite this article as: Slavich, G. M., & Cole, S. W. (2013). The emerging field of human social genomics. Clinical Psychological Science, 1, 331-348.
The Emerging Field of Human Social Genomics
George M. Slavich1 and Steven W. Cole1,2
1. Cousins Center for Psychoneuroimmunology and Department of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles
2. Department of Medicine, Division of Hematology-Oncology, UCLA Molecular Biology Institute, Jonsson Comprehensive Cancer Center, and UCLA AIDS Institute, University of California, Los Angele
Corresponding Author: George M. Slavich, Cousins Center for Psychoneuroimmunology, University of California, Los Angeles, UCLA Medical Plaza 300, Room 3156, Los Angeles, CA 90095-7076
Although we generally experience our bodies as being biologically stable across time and situations, an emerging field of research is demonstrating that external social conditions, especially our subjective perceptions of those conditions, can influence our most basic internal biological processes—namely, the expression of our genes. This research on human social genomics has begun to identify the types of genes that are subject to social-environmental regulation, the neural and molecular mechanisms that mediate the effects of social processes on gene expression, and the genetic polymorphisms that moderate individual differences in genomic sensitivity to social context. The molecular models resulting from this research provide new opportunities for understanding how social and genetic factors interact to shape complex behavioral phenotypes and susceptibility to disease. This research also sheds new light on the evolution of the human genome and challenges the fundamental belief that our molecular makeup is relatively stable and impermeable to social-environmental influence.
Keywords: stress, social isolation, social rejection, transcription factors, gene regulation, transcriptome, metagenomics, social epidemiology, 5-HTTLPR , RNA, DNA, interleukin-6, inflammation, disease, health
Research on the determinants of human health and behavior has long been dominated by the perspective that people are relatively stable biological entities. Underlying this view is the fact that we inherit the basic building blocks of human potential, DNA, from our parents and that our DNA remains largely unchanged over our lifetimes. The implications of this perspective have been profound. Because of such thinking, for example, we have framed years of scientific inquiry in terms of “genes by environment” and “nature versus nurture,” which imply that our genes are physiologically autonomous from the external social environment. These ideas have also shaped societal beliefs about the human body. Namely, although we are embedded in an ever-changing social world, we often experience ourselves as fundamentally separate from it: Genes influence behavior, we reckon, but not the other way around.
Although this perception of biological stability and impermeability is deeply engrained, an emerging field of research in human social genomics is beginning to prove these intuitions wrong. Contrary to the notion that our “molecular selves” are fixed across time and situations, for example, is increasing evidence that changes in the expression of literally hundreds of genes can occur as a function of the physical and social environments we inhabit. Moreover, it appears as though these effects are often more strongly tied to peoples’ subjective perceptions of their surrounding social environment (e.g., feeling lonely) than to “objective” features of those environments (e.g., being single). The human genome, therefore, is not a static blueprint for human potential. Instead, our genome appears to encode a wide variety of “potential biological selves,” and which “biological self” gets realized depends on the social conditions we experience over the life course.
Two recent discoveries have provided the empirical basis for these insights. First, functional genomics studies have shown that although DNA codes for the production of a wide variety of proteins that affect human functioning, our genetic code does not always result in the production of these proteins. Rather, it appears as though certain genes can be “turned on” and “turned off” by different social-environmental conditions. Second, research is now demonstrating that some social- environmental factors, such as social isolation and rejection, can influence the activity of not just a couple of genes but broad sets of hundreds of genes (i.e., gene profiles or gene programs). It has long been known that social-environmental conditions can shape complex behavioral phenotypes and susceptibility to disease (Cohen, Janicki-Deverts, & Miller, 2007). These new discoveries suggest that the external social world may exert these effects in part by influencing our most deeply internal biological processes—namely, the expression of our genes.
The goal of the present article is to review the rapidly developing literature on human social genomics, with a focus on the implications and possible future directions of this work. First, we provide a brief overview of human gene expression. Second, we review studies investigating the types of genes that are sensitive to external social conditions and the social conditions that appear to exert the strongest effects on gene expression. Third, we describe the neural and molecular mechanisms that mediate the effects of external social conditions on gene expression and discuss how these dynamics can cause social experiences to become biologically embedded. Fourth, we identify genetic factors that have been found to moderate individual differences in genomic sensitivity to social context. Finally, we outline several avenues for future clarification and research. The fact that social-environmental factors can cause broad changes in human gene expression represents a paradigm shift in research on Gene × Environment interplay, because it moves us away from thinking of these two factors as independent of each other. These discoveries also provide new insights into how the external social world gets not only “under the skin” but “onto the genome” to shape complex behavioral phenotypes and susceptibility to disease.
Experiences in daily life can come in many different forms, from feeling socially connected and physically safe to feeling socially isolated and rejected. Because we have no way of directly monitoring the molecular changes caused by these shifting social-environmental circumstances, we generally experience our bodies as being relatively stable over time and across the different situations that we encounter in everyday life. Deep inside our bodies, though, our cells are continually involved in a process of self-regeneration to replace dead cells and decayed proteins. In fact, because the average protein in the human body has a half-life of about 80 days (Welle, 1999), 1% to 2% of our entire molecular makeup must be replaced every day. As a result of this process, our physiological state on any given day can influence our molecular composition for weeks and months into the future.
The “genetic blueprint” for this biological regeneration is contained within the DNA of our approximately 21,000 genes. For this blueprint to affect human health and behavior, however, genes must be “turned on,” or expressed, in the form of RNA. Transcription of DNA into RNA is fundamentally regulated by intracellular proteins known as transcription factors. Some transcription factors are controlled endogenously within the cell to perpetuate its cellular identity. Other transcription factors act to alter gene expression in response to extracellular signals, such as hormones, neurotransmitters, or growth factors (Webster, Tonelli, & Sternberg, 2002; see Fig. 1). In the context of stress, for example, extracellular signals from glucocorticoids (e.g., cortisol) or catecholamines (e.g., norepinephrine) are detected by receptors on the surface of cells. Once detected, these signals initiate a complex set of intracellular interactions that result in the activation of transcription factors, such as the cyclic AMP response element-binding protein or the glucocorticoid receptor. This activation causes specific transcription factors to translocate into the nucleus of the cell where they bind to characteristic DNA motifs in gene promoters. Given that variations in the genetic code (i.e., DNA polymorphisms) can influence the affinity with which transcription factors bind, individual differences exist in the extent to which molecular signals are transduced. Assuming that binding has taken place, though, this process upregulates the transcription of DNA into mRNA, which is then decoded, or translated, to produce proteins that mediate bodily processes that have wide-ranging effects on mood, cognition, behavior, and health. Because such effects are ultimately determined by both the structure and the function of the genome, these dynamics have been likened to human brain function. In this analogy, DNA is like the structure of the brain insofar as it codes for an individuals’ biological and behavioral potential. For this potential to be realized, though, gene expression must occur—that is, DNA (i.e., the structure of the brain) must be transcribed into mRNA (i.e., functional brain activity) and then translated into protein.
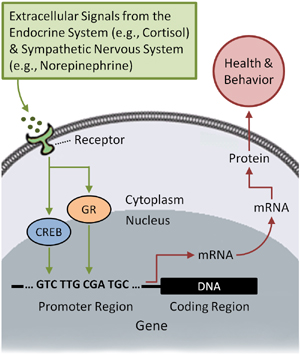
FIGURE 1. Exogenous control of human gene expression by stress. Central to human social genomics is the fact that social-environmental conditions, especially our subjective perceptions of those conditions, can reach deep inside the body to regulate the expression of broad sets of genes, or gene profiles. Receptors on the surface of cells “hear” extracellular signals from the endocrine and sympathetic nervous system, which respond to social experiences such as social isolation and rejection. Intracellular transcription factors, including the cyclic AMP response element-binding protein (CREB) and the glucocorticoid receptor (GR), then relay the signal to the nucleus of the cell, where the transcription factors bind to gene promoters and upregulate the transcription of DNA into mRNA. mRNA is then translated to produce amino acid sequences that form the basis for a wide range of proteins that influence human health and behavior. Individual differences in sensitivity to social context is influenced by the fact that several factors, such as a person’s genotype, can affect the binding of transcription factors to promoters, thereby influencing the likelihood that a particular signal results in gene transcription. Because only genes that are transcribed into RNA actually shape health outcomes and behavioral phenotypes, any process that influences transcription factor binding affinity (e.g., polymorphisms, methylation, histone modification) can substantially affect a person’s propensity to develop certain diseases or traits.
Exogenous control of human gene expression by stress. Central to human social genomics is the fact that social-environmental conditions, especially our subjective perceptions of those conditions, can reach deep inside the body to regulate the expression of broad sets of genes, or gene profiles. Receptors on the surface of cells “hear” extracellular signals from the endocrine and sympathetic nervous system, which respond to social experiences such as social isolation and rejection. Intracellular transcription factors, including the cyclic AMP response element-binding protein (CREB) and the glucocorticoid receptor (GR), then relay the signal to the nucleus of the cell, where the transcription factors bind to gene promoters and upregulate the transcription of DNA into mRNA. mRNA is then translated to produce amino acid sequences that form the basis for a wide range of proteins that influence human health and behavior. Individual differences in sensitivity to social context is influenced by the fact that several factors, such as a person’s genotype, can affect the binding of transcription factors to promoters, thereby influencing the likelihood that a particular signal results in gene transcription. Because only genes that are transcribed into RNA actually shape health outcomes and behavioral phenotypes, any process that influences transcription factor binding affinity (e.g., polymorphisms, methylation, histone modification) can substantially affect a person’s propensity to develop certain diseases or traits.
Gene expression occurs in all cell types and tissues, and which genes are expressed in a particular cell population at any given time is called the transcriptome. Scientists have long known that the basal transcriptome of different cell populations is guided by the evolved regulatory logic of the human genome and that the physicochemical environment plays an important role in shaping transcriptional dynamics (Gibson, 2008; Rappaport & Smith, 2010). More recently, though, evidence has accumulated demonstrating that social-environmental conditions, even purely imagined or symbolic cognitive representations of such conditions, can also trigger broad shifts in the basal transcriptome, thereby forming the empirical basis for human social genomics.
As we discuss later, social-environmental conditions influence basal transcriptome activity via central nervous system control of neural and endocrine processes (Cole, 2010; Irwin & Cole, 2011; McEwen, 2007; Miller, Chen, & Cole, 2009; Sterling, 2004). This ability for the central nervous system to regulate transcriptional dynamics is not accidental but rather part of a deep functional relationship between behavioral and biological responses that appear to have evolved to help protect individuals during times of physical threat (Cole, Hawkley, Arevalo, & Cacioppo, 2011; Irwin & Cole, 2011; Raison & Miller, 2013; Slavich, Way, Eisenberger, & Taylor, 2010). In fact, central nervous system regulation of human gene expression is critical for accelerating wound healing and limiting the spread of infection when physical injury has occurred. Because these pathways can be activated simply by subjective experiences of the world as hostile or threatening, however, genomewide transcriptional skewing that is imperative for surviving physical injury can also occur in the absence of actual physical danger, giving these dynamics the ability to affect risk for certain diseases (e.g., cardiovascular disease, cancer, neurodegenerative diseases, infectious diseases) in the contemporary social environment.
Early hints that social factors might significantly affect human gene expression came from studies assessing the influence that genetic versus environmental factors have on the basal transcriptome (Gibson, 2008; Idaghdour et al., 2010; Idaghdour, Storey, Jadallah, & Gibson, 2008). In a prototypic example, one study scanned the entire genome and found that whereas approximately 5% of differentially expressed genes could be attributed to genetic factors associated with gender and ancestry, more than 50% of differentially expressed genes were attributable to living in rural versus urban environments (Idaghdour et al., 2010). These studies could not disentangle the effects of physicochemical, microbial, and social influences on gene expression, but they did succeed in highlighting the previously unappreciated fact that environmental factors such as geographic location can regulate the expression of approximately 10 times as many genes as genetic factors.
Evidence that social environments may play a key role in such effects emerged around the same time from studies designed to examine the molecular basis for social influences on disease risk and longevity. For example, it has long been known that social isolation is associated with increased risk for mortality (Berkman & Kawachi, 2000; Holt-Lunstad, Smith, & Layton, 2010; House, 2001; House, Landis, & Umberson, 1988; Virchow, 1848/2006). To elucidate the biological mechanisms underlying these effects, early functional genomics studies recruited individuals experiencing chronic social isolation and persons who were socially well integrated. They then examined whether these two groups of individuals exhibited different genomewide transcriptional profiles. Because immune system functioning has important implications for health (Dhabhar, 2003; Glaser & Kiecolt-Glaser, 2005;Medzhitov, 2008), these studies focused on differential expression of two distinct modules of immune response genes—namely, proinflammatory cytokine genes (e.g.,IL1B, IL6, IL8, TNF), which combat bacteria and other extracellular pathogens, and antiviral immune response genes (e.g., IFNA, IFNB), which target intracellular pathogens such as viruses (Amit et al., 2009; Cole, 2012).
The first study conducted revealed a broad shift in the expression of two major gene programs within circulating immune cells from socially isolated versus socially integrated individuals (Cole et al., 2007). Compared to socially integrated individuals, people experiencing chronic social isolation showed enhanced expression of proinflammatory immune response genes and a reciprocal downregulation of antiviral immune response genes. These results were followed with bioinformatic analyses designed to reveal the molecular mechanisms mediating these transcriptional shifts. As expected, these analyses linked the genomewide transcriptional dynamics observed to increased activity of the transcription factors NF-κB and AP-1, which upregulate inflammation. The genomewide transcriptional shift also appeared to be driven by decreased activity of transcription factors such as interferon response factors, which promote innate antiviral resistance, and the glucocorticoid receptor, which plays a role in downregulating inflammation (Cole et al., 2007, 2011; for a review, see Hawkley, Cole, Capitanio, Norman, & Cacioppo, 2012). Because many of the diseases that socially isolated individuals develop involve heightened inflammatory activity (Glaser & Kiecolt-Glaser, 2005; Hawkley & Cacioppo, 2010; Heffner, Waring, Roberts, Eaton, & Gramling, 2011), these results provide a molecular basis for the elevated levels of morbidity and mortality that have long been observed in individuals experiencing loneliness and chronic social isolation.
Although this study focused on social isolation as a model of social-environmental adversity, similar proinflammatory/anti-antiviral skewing of the basal transcriptome in white blood cells (i.e., leukocytes) has emerged in people confronting other types of adversity as well. These adversities include acute social stressors, such as major life events involving targeted rejection (Murphy, Slavich, Rohleder, & Miller, 2013), as well as chronic social stressors, such as ongoing interpersonal difficulties (Miller, Rohleder, & Cole, 2009), imminent bereavement (Miller et al., 2008), and low socioeconomic status (Chen, Miller, Kobor, & Cole, 2011; Chen et al., 2009; Miller, Chen, Fok, et al., 2009). Consistent with the hypothesis that these effects have implications for health, proinflammatory/anti-antiviral skewing of the leukocyte basal transcriptome has also been found for individuals diagnosed with breast cancer (Antoni et al., 2012; Bower, Ganz, Irwin, Arevalo, & Cole, 2011) and posttraumatic stress disorder (O’Donovan et al., 2011).
Considered together, these correlational data suggest that immune response genes are highly sensitive to social-environmental conditions. This appears to be particularly true of immune response genes in “innate immune cells,” such as monocytes and dendritic cells, which mediate early responses to tissue damage. Converging evidence of such effects is provided by experimental research in mammalian model organisms (Cole et al., 2010; Cole et al., 2012; Sloan et al., 2010; Wohleb et al., 2011). In a recent study that manipulated the dominance rank (i.e., social status) of female macaques, for example, changes in rank over time caused widespread alterations in peripheral blood mononuclear cell gene expression (Tung et al., 2012). Specifically, macaques that were experimentally relegated to a lower social dominance rank exhibited upregulated expression of proinflammatory immune response genes and a related increase in glucocorticoid resistance, thus matching the previously described effects of social isolation on human gene expression. Moreover, these effects were associated with alterations in glucocorticoid signaling, suggesting that altered glucocorticoid functioning may play a key role in driving threat-related changes in proinflammatory gene expression. Finally, consistent with the hypothesis that these transcriptional alterations were due to the subordination manipulation, the mononuclear cell gene expression data alone predicted primates’ dominance rank with 80% accuracy (Tung et al., 2012).
In a more recent study (Cole et al., 2012), the genomewide transcriptional profiles of circulating leukocytes from infant macaques were analyzed for a group of rhesus macaques that were raised by their mother and for those raised by an inanimate surrogate mother or age-matched peers. Compared with peripheral blood mononuclear cells from maternally reared macaques, surrogate mother- and peer-reared macaques’ cells showed significantly greater expression of proinflammatory immune response genes and reduced expression of antiviral immune response genes. Consistent with the other human and animal model studies reviewed here, bioinformatic analyses designed to elucidate the mechanisms underlying these transcriptional shifts again implicated increased activity of proinflammatory NF-κB transcription factors and decreased activity of interferon response factor transcription factors in linking social adversity with leukocyte gene expression (Cole et al., 2012).
The pattern of upregulated proinflammatory immune response gene activity and down-regulated antiviral immune response gene activity described here represents what we call a conserved transcriptional response to adversity (CTRA). As illustrated in Figure 2, the CTRA generally involves a shift in the leukocyte’s basal transcriptional stance so that the body is better prepared to deal with different types of microbial exposure that have historically been associated with injury in dangerous environmental conditions (Cole et al., 2011; Irwin & Cole, 2011). These transcriptional shifts are critical in response to actual physical threat because they accelerate wound healing and limit infection (Irwin & Cole, 2011). In the very different health ecology of the contemporary social world (Finch, 2010; Gibson, 2009; McEwen, 2007; Richerson, Boyd, & Henrich, 2010; Sterling, 2004), however, chronic activation of the leukocyte CTRA in response to nonphysical (i.e., social) stressors contributes to a paradoxical increase in both (a) inflammation-related diseases such as cardiovascular disease and depression, caused by excessive proinflammatory immune response gene expression, and (b) vulnerability to viral infections such as the common cold, caused by insufficient antiviral immune response gene expression (Cole et al., 2007; Miller et al., 2008; seeMiller, Chen, & Cole, 2009). What once served as an adaptive response to impending physical threat now appears to be activated most frequently by the diverse array of actual or imagined social threats that humans experience on a daily basis.
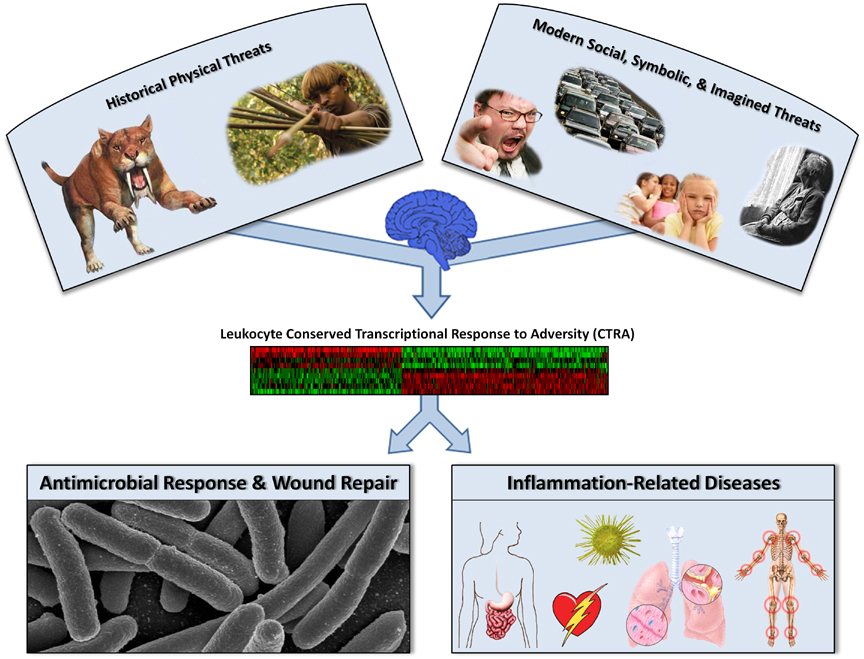
FIGURE 2. Conserved transcriptional response to adversity (CTRA). The CTRA emerges from neurobiological activation of leukocyte inflammatory genes and inhibition of innate antiviral genes in response to subjectively experienced physical or social threat. This proinflammatory skewing of the leukocyte basal transcriptome would be adaptive in response to physical threat, given that such threats were historically associated with increased risk for wounding and bacterial infection. However, social, symbolic, or imagined threats occurring in the contemporary social environment can also activate the CTRA, which maladaptively deflects host defenses away from the now more prevalent threat of socially mediated viral infections and toward the now diminished threats of injury and bacterial infection. Because the CTRA can be activated by imagined social threat (i.e., in the absence of actual physical threat), chronic activation of the CTRA can occur, which promotes the development of several inflammation-related conditions, including cardiovascular disease, depression, metabolic syndrome, neurodegenerative disorders, and certain neoplastic diseases. These psychiatric and physical conditions cause substantial morbidity and dominate modern mortality.
Conserved transcriptional response to adversity (CTRA). The CTRA emerges from neurobiological activation of leukocyte inflammatory genes and inhibition of innate antiviral genes in response to subjectively experienced physical or social threat. This proinflammatory skewing of the leukocyte basal transcriptome would be adaptive in response to physical threat, given that such threats were historically associated with increased risk for wounding and bacterial infection. However, social, symbolic, or imagined threats occurring in the contemporary social environment can also activate the CTRA, which maladaptively deflects host defenses away from the now more prevalent threat of socially mediated viral infections and toward the now diminished threats of injury and bacterial infection. Because the CTRA can be activated by imagined social threat (i.e., in the absence of actual physical threat), chronic activation of the CTRA can occur, which promotes the development of several inflammation-related conditions, including cardiovascular disease, depression, metabolic syndrome, neurodegenerative disorders, and certain neoplastic diseases. These psychiatric and physical conditions cause substantial morbidity and dominate modern mortality.
Gene programs such as the CTRA may have the ability to affect susceptibility to disease in part because of their sensitivity to social-environmental influences. For example, even transient environmental perturbations such as experimentally imposed sleep loss (Irwin, Wang, Campomayor, Collado-Hidalgo, & Cole, 2006), intense physical activity (Zieker et al., 2005), and acute psychological stress (Kawai et al., 2007; Nater et al., 2009) have been found to alter the leukocyte basal transcriptome. Social influences on gene expression can also penetrate surprisingly deeply into the body to remodel basal transcriptomes within several bodily tissues including the breast (Williams et al., 2009), lymph nodes (Sloan et al., 2007), and brain (Berton et al., 2006; Drnevich et al., 2012; Karssen et al., 2007; Weaver, Meaney, & Szyf, 2006). Similar effects have also been observed for diseased tissues, such as ovarian cancers (Lutgendorf et al., 2009), prostate cancers (Ornish et al., 2008), breast cancers (Sloan et al., 2010), and ischemic brain injuries (Karelina et al., 2009; see Cole, in press). Although additional research is needed to evaluate how widespread and deep the effects of social adversity on gene expression are, the fact that social influences can remodel transcriptional activity in the brain (and not just in the periphery of the body) is particularly important because it provides a biologically plausible explanation for how social adversity may elicit the wide range of neural, psychological, and behavioral alterations that characterize mental and physical health problems that have been associated with adverse social-environmental circumstances.
Given that several types of adversity (e.g., social isolation, subordination, rejection) have been found to influence gene expression, an important question to ask is, how exactly do external social conditions get converted into genome- regulating biochemistry? The mechanisms underlying this process—which we call social signal transduction—were first elucidated in animal model systems, such as zebra finch song learning, honeybee nurse/forager transition, and cichlid alpha male development (Robinson, Fernald, & Clayton, 2008). In each of these lines of research, analyses of socially induced transcriptional remodeling demonstrated a critical role for the central nervous system in transducing social information into changes in hormone and neurotransmitter dynamics that in turn modulate gene expression changes throughout the body, including in the brain (Fernald & Maruska, 2012; Robinson, Grozinger, & Whitfield, 2005; Robinson et al., 2008).
As illustrated in Figure 3a, human research has begun to identify broadly similar mechanisms, with central nervous system–mediated subjective perceptions of the social environment as safe versus threatening playing a key role in activating dynamics such as the leukocyte CTRA (Slavich, Way, et al., 2010). In studies of social isolation, for example, CTRA transcripts have been linked primarily to individuals’ subjective experience of social isolation or loneliness (i.e., a view of the social world as generally hostile and unsupportive; Cacioppo & Hawkley, 2009), rather than to “objective” parameters of social connection, such as individuals’ social network size or marital status (Cole et al., 2007; Cole et al., 2011). Analyses of socioeconomic-related differences in gene expression have revealed similar effects, with leukocyte transcriptional alterations being linked more closely to peoples’ general belief that the social world is a threatening or inhospitable place than to objective indices of social status, such as household income or education (Chen et al., 2009). Finally, as we have discussed already, even the mere threat of a significant social loss, such as the anticipated bereavement of a spouse with brain cancer, can activate the CTRA (Miller et al., 2008).
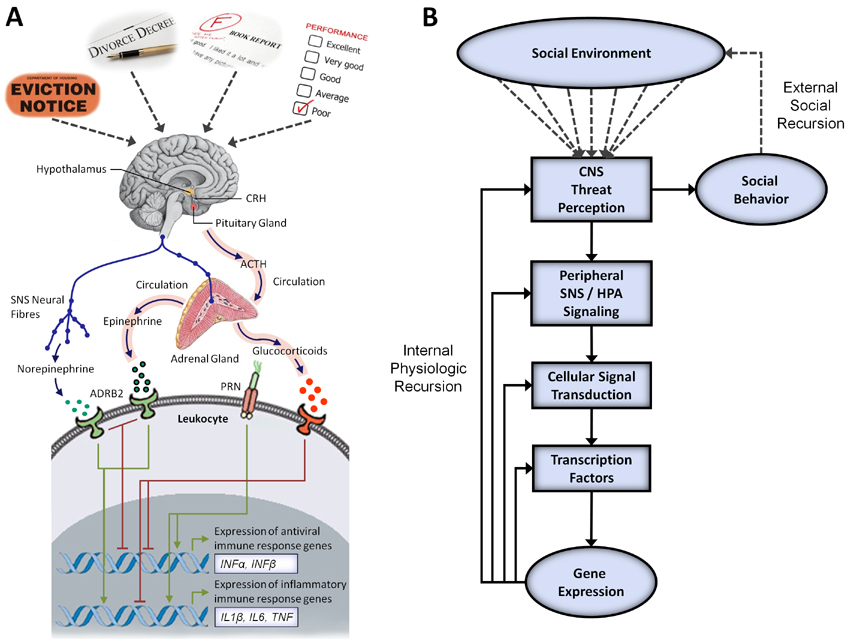
FIGURE 3. Human social signal transduction. Social signal transduction is the process by which subjectively perceived social conditions and historically and developmentally derived anticipatory worries alter genomewide transcriptional dynamics. (a) Social-environmental threats are neurocognitively appraised and converted into changing patterns of activity in the sympathetic nervous system (SNS) and hypothalamic-pituitary-adrenal (HPA) axis. Neuroeffector molecules from these systems engage specific gene transcriptional programs in differing target cells. In leukocytes, for example, SNS and HPA signaling suppress innate antiviral genes (e.g., IFNA, IFNB), whereas SNS signaling activates, and HPA signaling inhibits, proinflammatory cytokine genes (e.g., IL1B, IL6, IL8, TNF). (b) These processes can also be depicted conceptually, highlighting the fact that social experiences can become biologically embedded in at least two ways. First, internal physiologic recursion can occur, given that the genes targeted by social signal transduction pathways encode the molecules that mediate social signal transduction (e.g., receptors, intracellular signaling molecules, transcription factors, and growth factors). This recursive process propagates experienced-induced transcriptional alterations forward in time by sensitizing signal transduction pathways to the external social environment. Second, external social recursion can occur, given that social signal transduction can modulate genes involved in the regulation of social behavior (e.g., defensive responses to perceived threat). This recursive process takes place when conspecifics in the surrounding environment change their behavior in response to an individual’s altered actions, locking the individuals in a reciprocal feedback system. These two pathways give social-environmental experiences the ability to influence the basal cellular transcriptome for weeks and years after the initial environmental impetus has passed. ACTH = adrenocorticotropin hormone; ADRB2 = β2-adrenergic receptor; CRH = corticotrophin releasing hormone; PRR = pattern recognition receptor.
Human social signal transduction. Social signal transduction is the process by which subjectively perceived social conditions and historically and developmentally derived anticipatory worries alter genomewide transcriptional dynamics. (a) Social-environmental threats are neurocognitively appraised and converted into changing patterns of activity in the sympathetic nervous system (SNS) and hypothalamic-pituitary-adrenal (HPA) axis. Neuroeffector molecules from these systems engage specific gene transcriptional programs in differing target cells. In leukocytes, for example, SNS and HPA signaling suppress innate antiviral genes (e.g., IFNA, IFNB), whereas SNS signaling activates, and HPA signaling inhibits, proinflammatory cytokine genes (e.g., IL1B, IL6, IL8, TNF). (b) These processes can also be depicted conceptually, highlighting the fact that social experiences can become biologically embedded in at least two ways. First, internal physiologic recursion can occur, given that the genes targeted by social signal transduction pathways encode the molecules that mediate social signal transduction (e.g., receptors, intracellular signaling molecules, transcription factors, and growth factors). This recursive process propagates experienced-induced transcriptional alterations forward in time by sensitizing signal transduction pathways to the external social environment. Second, external social recursion can occur, given that social signal transduction can modulate genes involved in the regulation of social behavior (e.g., defensive responses to perceived threat). This recursive process takes place when conspecifics in the surrounding environment change their behavior in response to an individual’s altered actions, locking the individuals in a reciprocal feedback system. These two pathways give social-environmental experiences the ability to influence the basal cellular transcriptome for weeks and years after the initial environmental impetus has passed. ACTH = adrenocorticotropin hormone; ADRB2 = β2-adrenergic receptor; CRH = corticotrophin releasing hormone; PRR = pattern recognition receptor.
The paramount role of subjective perception in human social signal transduction stems from the fact that central nervous system–mediated experiences of social-environmental conditions, not the external conditions themselves, are what trigger the release of neural and endocrine response molecules that proximally regulate gene expression (Irwin & Cole, 2011; McEwen & Gianaros, 2010; Slavich, O’Donovan, Epel, & Kemeny, 2010; Slavich, Way, et al., 2010). Put another way, the effects of social experiences on gene expression are neurocognitively mediated. This has several implications. First, it means that purely symbolic or imagined stimuli—that is, situations that have not yet happened or that may never actually occur—can engage the same ancestral defense programs that are triggered by actual social or physical threats (see Fig. 2; Irwin & Cole, 2011; McEwen, 2007; Slavich, Way, et al., 2010;Sterling, 2004). Second, it suggests that the same social-environmental conditions, such as being socially evaluated or rejected, can be appraised in different ways and therefore have different gene expression consequences for different people depending on factors such as individuals’ sensitivity to social threat (Gyurak et al., 2012;O’Donovan, Slavich, Epel, & Neylan, 2013), propensity to view stressful circumstances as challenging versus threatening (Blascovich, Mendes, Hunter, & Salomon, 1999), cognitive-emotional resources (Taylor & Seeman, 1999), availability of social support (Eisenberger et al., 2011), and biographical and psychiatric histories (Slavich, Monroe, & Gotlib, 2011; Slavich, O’Donovan, et al., 2010). This explains in part how differences in perceptual processes and personality characteristics—such as negative cognitive biases, rejection sensitivity, social anxiety, and neuroticism—can come to be associated with stable individual differences in basal gene expression profiles and, therefore, health (Cole et al., 2007; Cole et al., 2011; Sloan, Capitanio, Tarara, & Cole, 2008). Finally, it indicates that events that are seemingly very different from one another, such as being socially rejected versus physically attacked, may have the ability to initiate the same transcriptional responses if they activate similar neurocognitive processes (e.g., given that social rejection and physical pain engage the same neural and endocrine pathways, both may induce proinflammatory skewing of the basal transcriptome; see Eisenberger, 2012; Eisenberger & Cole, 2012; Kross, Berman, Mischel, Smith, & Wager, 2011; Slavich, Way, et al., 2010).
One strategy for mapping the neural and hormonal signals that transduce experiences of the social environment into genome-regulating biochemistry involves using bioinformatic strategies that identify the types of intracellular transcription factors that mediate observed changes in gene expression. This research strategy has implicated several transcription control pathways in structuring genomewide transcriptional responses to social-environmental conditions. Some of these transcription factors are “usual suspects” that have long been known to be involved in threat-related neurobiology. A key set of transcription factors in this context are the cyclic AMP response element-binding protein family factors, which are activated by the β-adrenergic receptor (βAR)/protein kinase A signaling pathway during sympathetic nervous system-mediated fight-or-flight responses (Cole et al., 2007; Lutgendorf et al., 2009).
Other pathways involved in social signal transduction represent usual suspects behaving unusually. For example, although glucocorticoid receptors typically downregulate inflammatory gene expression, during exposure to chronic psychosocial stress or threat, glucocorticoid receptors fail to regulate glucocorticoid response genes, despite the fact that glucocorticoid levels are either normal or even elevated (Avitsur, Stark, Dhabhar, Kramer, & Sheridan, 2003; Miller et al., 2008; Wohleb et al., 2011). This phenomenon is called glucocorticoid insensitivity, and it involves βAR-induced increases in the production of immature leukocytes from bone marrow that bear functionally desensitized glucocorticoid receptor proteins (Cole et al., 2010;Pace, Hu, & Miller, 2007; Pace & Miller, 2009; Wohleb et al., 2011). As a result of decreased glucocorticoid receptor cross-regulation, NF-κB and AP-1 are de-repressed—that is, they are no longer inhibited from binding to gene promoters—thereby upregulating the proinflammatory component of the CTRA, leading to increased inflammatory activity (Cole et al., 2007; Pace, Hu, & Miller, 2011; Wohleb et al., 2011). At the same time, sympathetic nervous system/βAR signaling also inhibits interferon response factor transcription factors, which suppresses antiviral immune response gene expression and increases a person’s susceptibility to viral infection (Collado-Hidalgo, Sung, & Cole, 2006; Miller et al., 2008; Sloan et al., 2007; Sloan et al., 2010).
Several other mechanisms have been implicated in human social signal transduction. In addition to the transcription factors described so far, for example, there is evidence that EGR and GATA family transcription factors are involved in threat-related gene expression (Cole et al., 2007; Cole et al., 2010; Miller et al., 2008; Robinson et al., 2005, 2008). Whereas EGR transcription factors play a role in modulating key aspects of the adaptive immune response such as differentiation of lymphocyte precursors and activation of T cells and B cells (Gómez-Martín, Díaz-Zamudio, Galindo-Campos, & Alcocer-Varela, 2010), GATA transcription factors are expressed in a wide variety of tissues and regulate gene transcription in multiple stress-relevant organs, including the hypothalamus, pituitary, and adrenals (Viger, Guittot, Anttonen, Wilson, & Heikinheimo, 2008). In addition, mammalian animal model studies and postmortem studies of humans have demonstrated that adverse social experiences are associated with central nervous system alterations in other “epigenetic” processes, such as DNA methylation and histone modification, that could modulate gene transcriptional activity (Meaney & Ferguson-Smith, 2010; Robinson et al., 2005; Robinson et al., 2008;Tsankova, Renthal, Kumar, & Nestler, 2007). A landmark finding in this context is the discovery that poor maternal care in rodents (characterized by low licking/grooming and arched-back nursing; Weaver et al., 2004; Weaver et al., 2006) and childhood abuse in humans (characterized by a history of inappropriate sexual contact or severe physical abuse or neglect) are correlated with epigenetic modification of the glucocorticoid receptor gene in the neural transcriptome, particularly in the hypothalamus and amygdala (McGowan et al., 2009). These associations may thus help explain how adverse social-environmental experiences are translated into long-term changes in transcriptional activity that shape complex behavioral phenotypes and risk for disease (Boyce, Sokolowski, & Robinson, 2012; Fernald & Maruska, 2012).
Epigenetic processes represent one way by which adversities occurring at one point in time may lead to persistent changes in human behavior and disease risk. This issue is complex, though, and there are likely several nonmutually exclusive social-cognitive and biological processes that give social influences the ability to shape long-term behavior and health. One possibility (a social-environmental explanation) is that stressful situations may become prolonged. For example, a stressor might begin as an acute life event, such as the breakup of an intimate relationship, but then evolve into a chronic difficulty, such as social isolation and loneliness. A second possibility (a social-cognitive explanation) is that a stressor may be relatively acute in nature, but for some reason the person’s experience of the stressor becomes prolonged. For example, an individual is broken up with, and although the event has passed, he or she cannot stop thinking about the event and its meaning and implications (Nolen-Hoeksema, Wisco, & Lyubomirsky, 2008). Finally, a third possibility (a biological explanation) is that the stress exposure occurs during a developmentally critical or sensitive period, during which time transient social processes have the ability to reshape transcriptional dynamics and the biological systems that govern these dynamics (Miller & Chen, 2010; Miller, Chen, & Parker, 2011; Taylor, 2010).
The term generally used to describe this third process is biological embedding (Boyce et al., 2012; Hertzman, 1999; Hertzman & Boyce, 2010; Shonkoff, Boyce, & McEwen, 2009). One way that adverse experiences can get biologically embedded is by reshaping the dynamics of regenerative plasticity, or the ongoing low-level transcription of the human genome to replace dead cells and decayed proteins. In this process, increased hormone or neurotransmitter signaling caused by an experience of physical or social-environmental threat upregulates the ongoing production of new proteins or cells to replace those lost to damage or decay. One example of this process involves the hematopoietic output of leukocytes from bone marrow to replace the large numbers of white blood cells that die every day. Because leukocytes remain in the body for several days to years (depending on the exact cell type and location) and given that neurotransmitter regulation of hematopoietic output can selectively enhance the production of proinflammatory monocytes (Hanke, Powell, Stiner, Bailey, & Sheridan, 2012), experiences of social-environmental adversity occurring on any given day can reshape an individual’s inflammatory milieu well into the future (Cole et al., 2010; Irwin & Cole, 2011). Social influences can also induce short-term molecular alterations through transcriptional plasticity of tissue regeneration and wound-repair processes (Chen et al., 2011; Cole et al., 2007; Cole et al., 2010; Cole et al., 2011;Lutgendorf et al., 2009; Miller et al., 2008; Miller, Chen, Fok, et al., 2009; O’Donovan et al., 2011; Sloan et al., 2007; Sloan et al., 2010).
Social experiences can regulate gene expression in long-lived terminally differentiated cell types such as neurons as well (Robinson et al., 2005; Robinson et al., 2008;Sloan et al., 2008). For example, recent studies have found that both a general tendency to be unsociable and exposure to 3 weeks of social instability upregulate expression of the gene that codes for nerve growth factor beta (NGF) in rhesus macaque lymph nodes (Sloan et al., 2007; Sloan et al., 2008). This is critical because upregulated NGF expression has been associated with reduced antiviral immune response gene expression in leukocytes, which increases an organism’s vulnerability to viral infection (Collado-Hidalgo et al., 2006; Sloan et al., 2007). These dynamics have also been shown to trigger increased arborization of sympathetic nervous system fibers in the lymph node, which expands the regulatory pipeline from the brain to the immune system, making local leukocyte immune response gene and NGFtranscription increasingly sensitive to social-environmental input. Over time, this dynamic can become locked in a self-promoting positive feedback cycle that perpetuates sympathetic nervous system hyperinnervation, possibly leading to sustained subjective perceptions of heightened threat (Cole, 2012). A similar neuromolecular sensitization in the central nervous system may occur in response to chronic forms of stress, such as ongoing interpersonal problems, imminent bereavement, social isolation, and low socioeconomic status. As a result of such transcriptional recursion, which is depicted in Figure 3b, the molecular residue of experienced threat can become embedded in the basal cellular transcriptome and persist not just for days or months but for years after its initial environmental impetus has passed (Cole, 2012; Miller, Chen, Fok, et al., 2009; O’Donovan et al., 2011).
A major benefit of mechanistic models of social signal transduction is that they provide new opportunities for the systematic discovery of Gene × Environment interactions in human health and behavior. This can be done through computational modeling of the molecular processes that underlie Gene × Environment interactions. One way of approaching this is to first conduct bioinformatic analyses that identify transcriptional control pathways that are influenced by social- environmental input. Then, molecular interaction models can be used to analyze the effect that a particular genetic polymorphism has on the transcription factor’s ability to stimulate gene expression. If inflammatory processes are implicated, for example, analyses might focus on polymorphisms in the promoter of the interleukin-6 (i.e., IL6) gene, which regulates the production of the key proinflammatory cytokine IL-6 (Fishman et al., 1998). In contrast, if the serotonergic system is implicated, analyses might focus on polymorphisms in the promoter of the serotonin transporter gene (e.g., 5-HTTLPR), which has received considerable attention in studies of stress and health (Caspi et al., 2003; Caspi, Hariri, Holmes, Uher, & Moffitt, 2010). Finally, confirmation that a particular genetic polymorphism is relevant for a behavioral phenotype or health outcome can be achieved by testing whether the effects of a certain gene on the outcome of interest differ as a function of genotype.
Evidence of the benefits of this approach is provided by a recent study that elucidated the social signal transduction pathways underlying the effects of social-environmental adversity on mortality from inflammation-related causes of death (Cole et al., 2010). This study first identified the GATA1 transcription factor as a mediator of sympathetic nervous system/βAR-induced transcriptional responses to social- environmental threat. Next, predicted GATA1 target genes were scanned for polymorphisms that might affect the binding of GATA1 to characteristic DNA motifs. This analysis revealed a single nucleotide polymorphism—specifically, a G/C transversion 174 bases upstream of the transcription start site for the human IL6 gene—that was predicted to inhibit GATA1 binding. Under these circumstances, it was hypothesized that the IL6gene would effectively be “disconnected” from social-environmental regulation, thus preventing social adversity from “penetrating” the genome to impact health.
Consistent with this prediction, in vitro biochemical analyses confirmed that the C allele of the IL6 promoter showed reduced transcriptional responsiveness to βAR/GATA1 activation. In addition, in vivo molecular epidemiology confirmed that under high levels of social-environmental adversity, individuals who were homozygous for the GATA1-sensitive G allele died 2.8 years sooner than their counterparts bearing the GATA1-insensitive C allele. Consistent with the hypothesis that this effect was mediated by inflammatory factors, G allele carriers exhibited increased risk for inflammation-related causes of death (e.g., cardiovascular disease, cancer, diabetes) but not for noninflammation-related causes of death (e.g., respiratory failure, trauma). Moreover, the association between this polymorphism and mortality risk was completely attenuated when analyses adjusted for levels of systemic inflammation, as indexed by the inflammatory marker C-reactive protein (Cole et al., 2010). This effect is graphically illustrated in Figure 4.
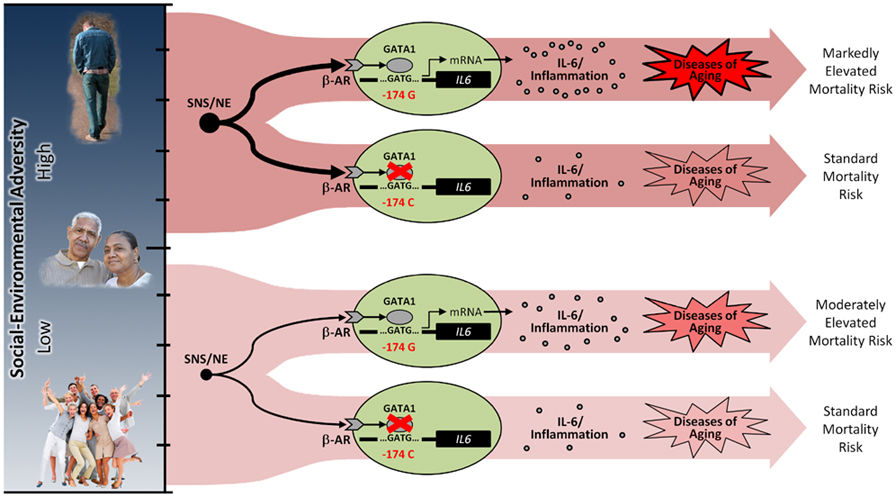
FIGURE 4. Concretizing gene-environment interactions in health. Graphically illustrated are the results of a recent study that showed that a single nucleotide polymorphism in the human IL6 promoter alters the ability for threat-activated GATA1 transcription factors to bind to DNA. Whereas high DNA binding affinity transduces the social threat–related signal downstream where it upregulatesIL6 gene expression, low binding affinity effectively disconnects this key proinflammatory cytokine from socioenvironmental regulation via sympathetic nervous system (SNS)/β-adrenergic receptor (βAR) signaling. Consistent with the prediction that these mechanisms are relevant for health, individuals who were homozygous for the GATA1-sensitive G allele died 2.8 years sooner than their counterparts bearing the GATA1-insensitive C allele. These results thus provide a genetic basis for differences in risk and resilience to social-environmental adversity, as well as a general framework for understanding why negative social experiences are more strongly related to increased IL6expressivity, systemic inflammation, and inflammation-related disease risk for some individuals compared to others. IL-6 = interleukin-6; NE = norepinephrine.
Concretizing gene-environment interactions in health. Graphically illustrated are the results of a recent study that showed that a single nucleotide polymorphism in the human IL6 promoter alters the ability for threat-activated GATA1 transcription factors to bind to DNA. Whereas high DNA binding affinity transduces the social threat–related signal downstream where it upregulatesIL6 gene expression, low binding affinity effectively disconnects this key proinflammatory cytokine from socioenvironmental regulation via sympathetic nervous system (SNS)/β-adrenergic receptor (βAR) signaling. Consistent with the prediction that these mechanisms are relevant for health, individuals who were homozygous for the GATA1-sensitive G allele died 2.8 years sooner than their counterparts bearing the GATA1-insensitive C allele. These results thus provide a genetic basis for differences in risk and resilience to social-environmental adversity, as well as a general framework for understanding why negative social experiences are more strongly related to increased IL6expressivity, systemic inflammation, and inflammation-related disease risk for some individuals compared to others. IL-6 = interleukin-6; NE = norepinephrine.
This social genomics approach to studying Gene × Environment interactions has several benefits for elucidating factors that influence human health. First, as would be expected, the approach reveals that for maximal phenotypic impact to occur, an individual has to have both an environmentally sensitive genotype (i.e., the IL6 G allele) and a social threat–related functional activation of this genotype, as indexed by sympathetic nervous system/βAR/GATA1 signaling. Second, this approach provides a concrete molecular basis for understanding why this IL6 polymorphism is not always associated with increased disease risk. Or, put another way, it identifies a genetic factor that explains individual differences in susceptibility to diseases that are caused in part by exposure to adverse social-environmental conditions. Finally, and perhaps most important, the approach helps map the social signal transduction pathway involved in inflammation-related disease in a way that highlights specific biological strategies, such as βAR blockade, that can be used for mitigating Gene × Environment-gated health risks (Cole et al., 2010; Sloan et al., 2010; Wohleb et al., 2011). For example, because inflammation has been implicated in several major disorders, including asthma, rheumatoid arthritis, cardiovascular disease, certain cancers, and depression, studies like the aforementioned could lead to novel methods for treating, and perhaps even preventing, some of our most common and burdensome diseases (Capuron & Miller, 2011; Haroon, Raison, & Miller, 2012; Hayley, 2011).
Put into a broader context, the Social Adversity × IL6 interaction described here exemplifies a growing trend in the discovery of polymorphisms that influence the magnitude of biological sensitivity to the environment, as opposed to phenotypes that are characterized by a specific directional risk (e.g., the “depression gene”) or benefit (e.g., the “altruism gene”; Boyce et al., 2012; Gibson, 2008; Way & Taylor, 2010). These phenotypic plasticity alleles confer increased advantage in favorable environments and increased risk in adverse environments, and they have likely remained polymorphic because they amplified a mix of fitness costs and benefits over the range of human evolutionary environments (Cole, 2012; Finch, 2010; Gibson, 2008,2009; Maranville et al., 2011; Richerson et al., 2010). Moreover, data increasingly suggest that plasticity alleles may be associated with a diverse array of phenotypes, including emotion perception and neural system activation, expression of distinct life history strategies involving reproductive and antisocial behavior, the development of human social networks, socioeconomic influences on depression, and the coevolution of genes and culture, all of which were discussed in a recent symposium on gene-environment interplay called “Genes & Environment: Finding the Missing Heritability of Complex Traits” (see http://www.gxe2010.org). Although it is unclear how broadly plasticity alleles affect human functioning, their existence raises the intriguing possibility that some people may be genetically more sensitive to transcriptional regulation by the social environment than others and thus more deeply enmeshed in an interlocking network of human genomes in which individual transcriptional “identities” are determined in part by the nature of the others surrounding them (whose own transcriptomes are in turn shaped by genomic regulation from others).
Despite these findings, plasticity alleles that enhance general adaptive capacities—such as the ability to anticipate challenges and preemptively deploy physiologic or behavioral defenses—would generally have “swept to fixation” over our evolutionary history (Gibson, 2008, 2009; McEwen, 2007; Sabeti et al., 2006; Sterling, 2004). As a result, much of the genetic basis for human transcriptional sensitivity to social context may now reside in nonpolymorphic (rather than polymorphic) regions of our genome. Because these genetic loci would have escaped detection in polymorphism-based association studies, it raises the interesting possibility that we may have yet to uncover the genetic sequences that most strongly shape biological sensitivity to social-environmental experiences (Gibson, 2008, 2009; Idaghdour et al., 2010).
Social influences regulate gene expression on an individual level, but they may also be involved in gene expression at a collective group level. In fact, social signal transduction implies a metagenomic system in which individual genomes operate differently depending on the presence of other people and their (subjectively perceived) implications for individual fitness outcomes, such as reproduction and survival (Cole et al., 2011; Slavich, Way, et al., 2010). The notion that the activity of our own genome is determined in part by the transcriptomes of others suggests the existence of a human metagenome—a system that is likely to generate complex emergent properties as transcriptional dynamics propagate through networks of genomes (Kauffman, 1993).
At this point in time, the existence of a human metagenomic system raises more questions than answers. For example, which types of genes are subject to network-level regulation (Cole et al., 2011; Robinson et al., 2008; Slavich, Way, et al., 2010)? How are network transcriptional dynamics affected by genetic, historical, or developmental influences or by network structural characteristics such as linkage patterns, community blocks, and individual linkage characteristics including centrality, density, or redundancy? Do individual transcriptional alterations affect network structure—for example, the tendency to affiliate with similar others (i.e., homophily) or to bond with those who are different (i.e., heterophily)? How do physicochemical or microbial factors interact with human social systems to regulate metagenomic systems (Gibson, 2008), and are these influences transmitted through different networks or signal transduction pathways than social threat–related influences? Objective and subjective network characteristics regulate distinct groups of immune response genes (Cole et al., 2007; Cole et al., 2011), as well as neural-regulatory genes (Berton et al., 2006; Robinson et al., 2008; Weaver et al., 2006), but their effects on other types of genes and bodily tissues remain poorly understood. Furthermore, what role does human culture play in metagenomic dynamics (Boyd, Richerson, & Henrich, 2011; Richerson et al., 2010)? Finally, what network features prompt our ancient hunter-gatherer genomes to deploy transcriptional host defense programs such as the CTRA in modern social ecologies (Finch, 2007, 2010; McEwen, 2007; Richerson et al., 2010; Slavich, Way, et al., 2010; Sterling, 2004)? Activating a transcriptional response that reduces risk of injury and infection during actual physical danger makes good adaptive sense; when such a response is deployed during contemporary social stressors, however, the health risks can easily outweigh the benefits, particularly if the response is prolonged by neurocognitive mechanisms or transcriptional embedding.
These questions begin to highlight what remains to be discovered within the exciting new field of human social genomics. Many other issues await further investigation. In our discussion of socially sensitive genes, for example, we focused on gene programs involved in innate immunity and, specifically, inflammation, given the adaptive importance of these genes and the central role that they play in promoting susceptibility to a variety of diseases. However, what other types of genes are responsive to social input? Furthermore, any social- psychological process or complex phenotype that has evolutionary significance could presumably be represented at the level of the genome or metagenome. These include social attachment, affiliation, altruism, conformity, personality, psychopathology, prejudice, social hierarchies, mating behavior, and culture. Another major avenue for future discovery, therefore, is to identify additional complex phenotypes that are influenced by social context and mediated by transcriptional changes. This research should aim to specify the gene programs involved in these phenotypes, the physiological pathways that mediate the effects of social processes on these transcriptional dynamics, and the implications that these processes have for the behavioral or health outcomes under investigation.
Relevant for the identification of socially sensitive genes is the related question of what types of social-environmental stressors are most strongly related to changes in gene expression. In the present discussion, we focused on particular types of experiences (e.g., low social status, social isolation, rejection) and social-environmental conditions (e.g., living in a rural vs. urban environment). Our perspective is that leukocyte transcriptional dynamics have evolved to counter the differing types of microbial exposure that have historically been associated with friendly versus threatening conditions. But, presumably other types of contemporary social-environmental stressors or experiences could indicate a possible increase in threat and therefore have implications for human gene expression. So, what, exactly, are the types of stressors or social-environmental experiences that are most strongly related to gene expression and, in turn, to different health outcomes and behavioral phenotypes (Cole, 2012; Harkness et al., 2010; Kemeny, 2009; Lutgendorf et al., 2012; Monroe & Slavich, 2007; Monroe, Slavich, & Georgiades, 2009; Slavich, Thornton, Torres, Monroe, & Gotlib, 2009)? And, are the mechanisms that link these different experiences to transcriptional changes the same or different for different social influences (O’Donovan et al., 2010)?
Future research is also needed to elucidate the neurocognitive processes that are relevant for gene expression. In the present review, we summarized data showing that subjective perceptions of the external social environment (e.g., as being friendly vs. hostile) appear to be more strongly related to genomewide transcriptional shifts than the actual social-environmental conditions themselves. This does not mean that the contextual features and characteristics of the external social world do not matter for gene expression, especially since peoples’ subjective experiences are shaped in part by the reality of their surrounding social environment. But, it does suggest that subjective neurocognitive appraisals of the external social environment are important for social threat–related changes in gene expression.
To our knowledge, only one study to date has examined how neurocognitive perceptions of the external social world relate to inflammatory biology, and this study found that individuals who exhibited greater neural responses to social rejection in pain-related neural regions—specifically, the dorsal anterior cingulate cortex and bilateral anterior insula—had greater inflammatory responses to a laboratory-based social stress task (Slavich, Way, et al., 2010). These findings suggest that the neural substrates of inflammation-related gene expression may include brain systems that are involved in processing experiences of physical pain (Eisenberger, 2012; Eisenberger & Cole, 2012). However, several neural systems are likely relevant for gene expression. The system just described, for example, appears to be involved in the experience of social-environmental threat and includes the amygdala, dorsal anterior cingulate cortex, and bilateral anterior insula. In addition, a second system appears to be implicated in determining the relative physical safety and security of the individual and is mediated in part by the ventromedial prefrontal cortex. Finally, a third system is involved in representing the thoughts and intentions of others and their relevance for the self (e.g., “What do those people think of me?” or “Do they like me?”) and includes the dorsomedial prefrontal cortex, medial prefrontal cortex, pregenual anterior cingulate, and posterior cingulate (Eisenberger et al., 2011; Muscatell et al., 2012). Because no studies have examined the neurocognitive processes associated with changes in gene expression, an important goal for future research is to elucidate the neural systems that regulate transcriptional dynamics and to determine how these neuromolecular interactions are relevant for human health and behavior (for a more extensive review of these neural systems and how they may influence gene expression, see Irwin & Cole, 2011; Lieberman, 2010; Muscatell & Eisenberger, 2012).
In addition to identifying the neurocognitive processes that are relevant for gene expression, research is needed to elucidate the full array of mechanisms that influence whether neurocognitive signals get converted into genomewide transcriptional events. For example, genetic polymorphisms affect social signal transduction, but researchers in the social and health sciences have focused almost exclusively on a very small number of polymorphisms (e.g., 5-HTTLPR, IL6 promoter) that may be relevant for human health and behavior. Additional research is thus needed to examine other polymorphisms in both regulatory and coding regions of DNA that may be important for gene expression. In addition, although we have discussed how translated regions of mRNA regulate gene expression, untranslated regions of mRNA also play an important role in controlling gene expression changes insofar as they influence mRNA maturation, shuttling, stability, and translational efficiency (Asson-Batres, Spurgeon, Diaz, DeLoughery, & Bagby, 1994; Stamou & Kontoyiannis, 2010). Put broadly, then, there is still much to learn about the full range of mechanisms that influence social signal transduction.
Important questions also remain concerning how social environmentally induced changes in gene expression have long-term effects. Because our cells are continually involved in a process of self-regeneration, experiences occurring at any point in time can influence human functioning for weeks through regenerative plasticity and for months and years via neuromolecular sensitization (Cole, 2009; Irwin & Cole, 2011). Initial evidence for such sensitization is suggested by research showing that neural activity can regulate inflammatory processes (Slavich, Way, et al., 2010) and that changes in inflammation can in turn influence neural activity (Eisenberger, Inagaki, Rameson, Mashal, & Irwin, 2009). As we have already noted, though, no studies have directly examined the neural processes that regulate gene expression changes (and vice versa) in humans. So, what are the mechanisms that underlie these effects, and what implications do these dynamics have for different types of behavioral or health outcomes? In addition, can these dynamics help explain how some behavioral routines or addictions develop, why psychiatric disorders like anxiety and depression are often recurrent, or why certain types of psychological experiences (e.g., anxiety, depression) and somatic conditions (e.g., asthma, cardiovascular disease) frequently co-occur? Although these bidirectional links have possible implications for understanding how adverse experiences become biologically embedded and thus influence health and behavior for years after a social-environmental perturbation has passed, much more research is needed to determine how these effects occur.
Most of what we have said so far suggests that only adverse social-environmental conditions influence gene expression, leading to negative health outcomes and behaviors. This is certainly not the case, though, and in the long run, the most impactful human social genomics studies may well be those that identify the types of positive experiences that influence gene expression. In one recent study, for example, a 10-week cognitive-behavior intervention designed to target anxiety-related affective and behavioral processes counteracted the effects of stress-related CTRA transcriptional skewing by reducing expression of proinflammatory and metastasis-related genes and by increasing expression of interferon-related genes in early-stage breast cancer patients who were randomly assigned to receive the intervention (Antoni et al., 2012). Similar genome-regulating effects have been found for Kirtan Kriya meditation (Black et al., in press) and for a mindfulness-based stress reduction program (Creswell et al., 2012). These data thus provide the first evidence that psychological interventions can reverse stress-induced genomewide transcriptional responses such as the CTRA, which may in turn have implications for human health (Finch, 2007, 2010).
Such findings raise the question of what other cognitive or behavioral interventions might affect the basal transcriptome and, when identified, whether these interventions can be deployed to improve clinical outcomes and quality of life—and to reduce suffering and disease burden—in major diseases such as cardiovascular disease, depression, and cancer. This research also hints at the possibility that certain positive states of mind may be associated with differences in gene expression, which may in turn shape a person’s risk for a variety of psychiatric and physical disorders. Such states may include optimism, tranquility, affection, gratitude, admiration, mindfulness, social connectedness, and compassion (Aschbacher et al., 2012; Eisenberger et al., 2011; Epel et al., 2013; Immordino-Yang, McColl, Damasio, & Damasio, 2009;Keltner, Marsh, & Smith, 2010). However, because these states generally involve affiliative, appetitive, exploratory, and reward motivations (e.g., instead of isolative and avoidant motivations), they likely engage neurobiological pathways and influence sets of genes that are different from those affected during physical or social threat (Lieberman & Eisenberger, 2009; Panksepp, 1998). Clearly, we are just beginning to understand how genomic activity differs as a function of the full range of positive and negative social conditions that humans experience.
Finally, many promising avenues of research exist relating to how social-environmental factors may regulate gene expression dynamics on a collective level. For example, it is known that individuals from different geographic regions and races have different polymorphic structures, but what are the implications of this genetic variation for basal transcriptome differences across social groups, cultures, and countries? Can understanding these genetic and genomic differences help explain why certain diseases and phenotypes cluster in particular groups or populations? In addition, what role does human culture play in social signal transduction, and how has social signal transduction shaped the evolution of the human genome, the development of human social systems, and gene-culture coevolution (Richerson et al., 2010)? These questions highlight just a few of the many exciting ways in which integrating human social genomics and social systems analyses can advance our understanding of contemporary human biology to illuminate the deeper evolutionary history of Homo sapiens, the most social of all animals (Cacioppo & Hawkley, 2009).
In conclusion, the long-standing belief in science and society has been that we are relatively stable biological entities, despite the fact that we live in a dynamic social environment. From this perspective, it has made sense to ask questions that pit genes against environment and nature against nurture. As we learn more about human gene transcription, however, it is becoming increasingly apparent that our molecular “selves” are far more fluid and permeable to social-environmental influence than we have generally appreciated (Robinson, 2004). To capture the essence of the rapidly developing literature on this topic, we have called this field of research human social genomics and have reviewed the empirical studies that substantiate its existence, relevance, and importance.
As we have summarized here, this burgeoning field of research has begun to identify the specific types of genes that are subject to social-environmental regulation, the neural and molecular mechanisms that mediate the effects social processes have on gene expression, and the genetic polymorphisms that moderate the extent to which social conditions cause transcriptional shifts. The discovery that social-environmental factors can substantially alter the expression of literally hundreds of genes and that the types of genes that are differentially expressed can be meaningfully identified as gene programs or profiles represents a paradigm shift in thinking on gene-environment interactions. It also provides new insights into how social conditions shape complex phenotypes and susceptibility to disease and, in doing so, highlights potential targets for mitigating the negative effects of these phenomena. Finally, these results provide new theoretical and computational modeling frameworks that link human molecular genetics to genomic evolution and gene-culture coevolution. For although as adults we are often physically separated from those around us, our presence in different social groups means that we are transcriptionally connected, giving rise to a human metagenome that has implications for collective health and behavior.
Looking forward, several broad avenues exist for additional research and discovery. First, since most of the existing studies on human social genomics are correlational, additional human and animal model studies are needed in which transcriptional changes are examined following exposure to experimentally manipulated social-environmental conditions. These studies will expand the empirical literature on human social genomics and reveal new insights into the types of social-environmental conditions that are most strongly associated with genomewide transcriptional shifts. Second, to better map human social signal transduction pathways, studies will need to assess multiple levels of analysis simultaneously. For example, by employing measures of neural activity, gene expression, and psychological and behavioral processes in the same experimental context, researchers will be able to elucidate the neural systems that regulate genomewide transcriptional dynamics and the psychological and behavioral correlates of these dynamics, all linked to the same social-environmental provocation. Finally, additional studies are needed to examine the relevance of social genomics research for different psychiatric and physical diseases and to translate findings from this work into new psychological and psychopharmacological strategies for reducing the morbidity and mortality associated with our most common and costly disorders. Studies that elucidate how the external social environment gets translated into the internal biological environment of disease pathogenesis may lead to new methods for preventing disorders and promoting wellness, but much more research is needed to turn this promise into reality.
Declaration of Conflicting Interests: The authors declare that they have no conflicts of interest with respect to their authorship or the publication of this article.
Funding: Preparation of this review was supported by a Society in Science–Branco Weiss Fellowship to George Slavich, and by National Institutes of Health grants R01-CA116778, R01-AG033590, and P30-AG028748 to Steven Cole.
Received July 20, 2012
Accepted January 23, 2013
References
Amit, I., Garber, M., Chevrier, N., Leite, A. P., Donner, Y., Eisenhaure, T., Guttman, M., Grenier, J. K., Li, W., Zuk, O., Schubert, L. A., Birditt, B., Shay, T., Goren, A., Zhang, X., Smith, Z., Deering, R., McDonald, R. C., Cabili, M., Bernstein, B. E., Rinn, J. L., Meissner, A., Root, D. E., Hacohen, N., & Regev, A. (2009). Unbiased reconstruction of a mammalian transcriptional network mediating pathogen responses. Science, 326, 257–263.
Antoni, M. H., Lutgendorf, S. K., Blomberg, B., Carver, C. S., Lechner, S., Diaz, A., Stagl, J., Arevalo, J. M., & Cole, S. W. (2012). Cognitive-behavioral stress management reverses anxiety-related leukocyte transcriptional dynamics. Biological Psychiatry, 71, 366–372.
Aschbacher, K., Epel, E., Wolkowitz, O. M., Prather, A. A., Puterman, E., & Dhabhar, F. S. (2012). Maintenance of a positive outlook during acute stress protects against pro-inflammatory reactivity and future depressive symptoms. Brain, Behavior, and Immunity, 26, 346–352.
Asson-Batres, M. A., Spurgeon, S. L., Diaz, J., DeLoughery, T. G., Bagby, G. C., Jr. (1994). Evolutionary conservation of the AU-rich 3’ untranslated region of messenger RNA. Proceedings of the National Academy of Sciences of the United States of America, 91, 1318–1322.
Avitsur, R., Stark, J. L., Dhabhar, F. S., Kramer, K. A., & Sheridan, J. F. (2003). Social experience alters the response to social stress in mice. Brain, Behavior, and Immunity, 17, 426–437.
Berkman, L. F., & Kawachi, I. (2000). Social epidemiology. New York: Oxford University Press.
Berton, O., McClung, C. A., Dileone, R. J., Krishnan, V., Renthal, W., Russo, S. J., Graham, D., Tsankova, N. M., Bolanos, C. A., Rios, M., Monteggia, L. M., Self, D. W., & Nestler, E. J. (2006). Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science, 311, 864–868.
Black, D. S., Cole, S. W., Irwin, M. R., Breen, E., St Cyr, N. M., Nazarian, N., Khalsa, D. S., & Lavretsky, H. (in press). Yogic meditation reverses NF-κB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology.
Blascovich, J., Mendes, W. B., Hunter, S. B., & Salomon, K. (1999). Social “facilitation” as challenge and threat. Journal of Personality and Social Psychology, 77, 68–77.
Bower, J. E., Ganz, P. A., Irwin, M. R., Arevalo, J. M., & Cole, S. W. (2011). Fatigue and gene expression in human leukocytes: Increased NF-κB and decreased glucocorticoid signaling in breast cancer survivors with persistent fatigue. Brain, Behavior, and Immunity, 25, 147–150.
Boyce, W. T., Sokolowski, M. B., & Robinson, G. E. (2012). Toward a new biology of social adversity. Proceedings of the National Academy of Sciences of the United States of America, 109, 17143–17148.
Boyd, R., Richerson, P. J., & Henrich, J. (2011). The cultural niche: Why social learning is essential for human adaptation. Proceedings of the National Academy of Sciences of the United States of America, 108, 10918–10925.
Cacioppo, J. T., & Hawkley, L. C. (2009). Perceived social isolation and cognition. Trends in Cognitive Sciences, 13, 447–454.
Capuron, L., & Miller, A. H. (2011). Immune system to brain signaling: Neuropsychopharmacological implications. Pharmacology and Therapeutics, 130, 226–238.
Caspi, A., Hariri, A. R., Holmes, A., Uher, R., & Moffitt, T. E. (2010). Genetic sensitivity to the environment: The case of the serotonin transporter gene and its implications for studying complex diseases and traits. American Journal of Psychiatry, 167, 509–527.
Caspi, A., Sugden, K., Moffitt, T. E., Taylor, A., Craig, I. W., Harrington, H., McClay, J., Mill, J., Martin, J., Braithwaite, A., & Poulton, R. (2003). Influence of life stress on depression: Moderation by a polymorphism in the 5-HTT gene. Science, 301, 386–389.
Chen, E., Miller, G. E., Kobor, M. S., & Cole, S. W. (2011). Maternal warmth buffers the effects of low early-life socioeconomic status on pro-inflammatory signaling in adulthood. Molecular Psychiatry, 16, 729–737.
Chen, E., Miller, G. E., Walker, H. A., Arevalo, J. M., Sung, C. Y., & Cole, S. W. (2009). Genome-wide transcriptional profiling linked to social class in asthma. Thorax, 64, 38–43.
Cohen, S., Janicki-Deverts, D., & Miller, G. E. (2007). Psychological stress and disease. Journal of the American Medical Association, 298, 1685–1687.
Cole, S. W. (2009). Social regulation of human gene expression. Current Directions in Psychological Science, 18, 132–137.
Cole, S. W. (2010). Elevating the perspective on human stress genomics. Psychoneuroendocrinology, 35, 955–962.
Cole, S. W. (2012). Social regulation of gene expression in the immune system. In S. C. Segerstrom (Ed.), The Oxford Handbook of Psychoneuroimmunology (pp. 254–273). New York: Oxford University Press.
Cole, S. W. (in press). Nervous system regulation of the cancer genome. Brain, Behavior, and Immunity.
Cole, S. W., Arevalo, J. M., Takahashi, R., Sloan, E. K., Lutgendorf, S. K., Sood, A. K., Sheridan, J. F., & Seeman, T. E. (2010). Computational identification of gene-social environment interaction at the human IL6 locus. Proceedings of the National Academy of Sciences of the United States of America, 107, 5681–5686.
Cole, S. W., Conti, G., Arevalo, J. M., Ruggiero, A. M., Heckman, J. J., & Suomi, S. J. (in press). Transcriptional modulation of the developing immune system by early life social adversity. Proceedings of the National Academy of Sciences of the United States of America.
Cole, S. W., Hawkley, L. C., Arevalo, J. M., & Cacioppo, J. T. (2011). Transcript origin analysis identifies antigen-presenting cells as primary targets of socially regulated gene expression in leukocytes. Proceedings of the National Academy of Sciences of the United States of America, 108, 3080–3085.
Cole, S. W., Hawkley, L. C., Arevalo, J. M., Sung, C. Y., Rose, R. M., & Cacioppo, J. T. (2007). Social regulation of gene expression in human leukocytes. Genome Biology, 8, R189.
Collado-Hidalgo, A., Sung, C., & Cole, S. (2006). Adrenergic inhibition of innate anti-viral response: PKA blockade of Type I interferon gene transcription mediates catecholamine support for HIV-1 replication. Brain, Behavior, and Immunity, 20, 552–563.
Creswell, J. D., Irwin, M. R., Burklund, L. J., Lieberman, M. D., Arevalo, J. M., Ma, J., Breen, E. C., & Cole, S. W. (2012). Mindfulness-Based Stress Reduction training reduces loneliness and pro-inflammatory gene expression in older adults: A small randomized controlled trial. Brain, Behavior, and Immunity, 26, 1095–1101.
Dhabhar, F. S. (2003). Stress, leukocyte trafficking, and the augmentation of skin immune function. Annals of the New York Academy of Sciences, 992, 205–217.
Drnevich, J., Replogle, K. L., Lovell, P., Hahn, T. P., Johnson, F., Mast, T. G., Nordeen, E., Nordeen, K., Strand, C., London, S. E., Mukai, M., Wingfield, J. C., Arnold, A. P., Ball, G. F., Brenowitz, E. A., Wade, J., Mello, C. V., & Clayton, D. F. (2012). Impact of experience-dependent and -independent factors on gene expression in songbird brain. Proceedings of the National Academy of Sciences of the United States of America, 109, 17245–17252.
Eisenberger, N. I. (2012). The pain of social disconnection: Examining the shared neural underpinnings of physical and social pain. Nature Reviews Neuroscience, 13, 421–434.
Eisenberger, N. I., & Cole, S. W. (2012). Social neuroscience and health: Neurophysiological mechanisms linking social ties with physical health. Nature Neuroscience, 15, 669–674.
Eisenberger, N. I., Inagaki, T. K., Rameson, L. T., Mashal, N. M., & Irwin, M. R. (2009). An fMRI study of cytokine-induced depressed mood and social pain: The role of sex differences. Neuroimage, 47, 881–890.
Eisenberger, N. I., Master, S. L., Inagaki, T. K., Taylor S. E., Shirinyan, D., Lieberman, M. D., Naliboff, B. D. (2011). Attachment figures activate a safety signal-related neural region and reduce pain experience. Proceedings of the National Academy of Sciences of the United States of America, 108, 11721–11726.
Epel, E. S., Puterman, E., Lin, J., Blackburn, E., Lazaro, A, & Mendes, W. B. (2013). Wandering minds and aging cells. Clinical Psychological Science, 1, 75–83.
Fernald, R. D., & Maruska, K. P. Social information changes the brain. Proceedings of the National Academy of Sciences of the United States of America, 109, 17194–17199.
Finch, C. E. (2007). The biology of human longevity: Inflammation, nutrition, and aging in the evolution of lifespans. Burlington, M.A.: Academic Press.
Finch, C. E. (2010). Evolution of the human lifespan and diseases of aging: Roles of infection, inflammation, and nutrition. Proceedings of the National Academy of Sciences of the United States of America, 107, 1718–1724.
Fishman, D., Faulds, G., Jeffery, R., Mohamed-Ali, V., Yudkin, J. S., Humphries, S., & Woo, P. (1998). The effect of novel polymorphisms in the interleukin-6 (IL-6) gene on IL-6 transcription and plasma IL-6 levels, and an association with systemic-onset juvenile chronic arthritis. Journal of Clinical Investigation, 102, 1369–1376.
Gibson, G. (2008). The environmental contribution to gene expression profiles. Nature Reviews Genetics, 9, 575–581.
Gibson, G. (2009). Decanalization and the origin of complex disease. Nature Reviews Genetics, 10, 134–140.
Glaser, R., & Kiecolt-Glaser, J. K. (2005). Stress-induced immune dysfunction: Implications for health. Nature Reviews Immunology, 5, 243–251.
Gómez-Martín, D., Díaz-Zamudio, M., Galindo-Campos, M., & Alcocer-Varela, J. (2010). Early growth response transcription factors and the modulation of immune response: Implications towards autoimmunity. Autoimmune Reviews, 9, 454–458.
Gyurak, A., Hooker, C. I., Miyakawa, A., Verosky, S., Luerssen, A., & Ayduk, Ö. N. (2012). Individual differences in neural responses to social rejection: The joint effect of self-esteem and attentional control. Social, Cognitive, and Affective Neuroscience, 7, 322–331.
Hanke, M. L., Powell, N. D., Stiner, L. M., Bailey, M. T., & Sheridan, J. F. (2012). Beta adrenergic blockade decreases the immunomodulatory effects of social disruption stress. Brain, Behavior, and Immunity, 26, 1150–1159.
Harkness, K. L., Tabari, N., Monroe, S. M., Slavich, G. M., Gotlib, I. H., & Bagby, R. M. (2010). Sex differences in life events prior to onset of major depressive disorder: the moderating effect of age. Journal of Abnormal Psychology, 119, 791–803.
Haroon, E., Raison, C. L., & Miller, A. H. (2012). Psychoneuroimmunology meets neuropsychopharmacology: Translational implications of the impact of inflammation on behavior. Neuropsychopharmacology, 37, 137–162.
Hawkley, L. C., & Cacioppo, J. T. (2010). Loneliness matters: A theoretical and empirical review of consequences and mechanisms. Annals of Behavioral Medicine, 40, 218–227.
Hawkley, L. C., Cole, S. W., Capitanio, J. P., Norman, G. J., & Cacioppo, J. T. (2012). Effects of social isolation on glucocorticoid regulation in social mammals. Hormones and Behavior, 62, 314–323.
Hayley, S. (2011). Toward an anti-inflammatory strategy for depression. Frontiers in Behavioral Neuroscience, 5, 19.
Heffner, K. L., Waring, M. E., Roberts, M. B., Eaton, C. B., & Gramling, R. (2011). Social isolation, C-reactive protein, and coronary heart disease mortality among community-dwelling adults. Social Sciences and Medicine, 72, 1482–1488.
Hertzman, C. (1999). The biological embedding of early experience and its effects on health in adulthood. Annals of the New York Academy of Sciences, 896, 85–95.
Hertzman, C., & Boyce, T. (2010). How experience gets under the skin to create gradients in developmental health. Annual Review of Public Health, 31, 329–347.
Holt-Lunstad, J., Smith, T. B., & Layton, J. B. (2010). Social relationships and mortality risk: A meta-analytic review. PLoS Medicine, 7, e1000316.
House, J. S. (2001). Social isolation kills, but how and why? Psychosomatic Medicine, 63, 273–274.
House, J. S., Landis, K. R., & Umberson, D. (1988). Social relationships and health. Science, 241, 540–545.
Idaghdour, Y., Czika, W., Shianna, K. V., Lee, S. H., Visscher, P. M., Martin, H. C., Miclaus, K., Jadallah, S. J., Goldstein, D. B., Wolfinger, R. D., & Gibson, G. (2010). Geographical genomics of human leukocyte gene expression variation in southern Morocco. Nature Genetics, 42, 62–67.
Idaghdour, Y., Storey, J. D., Jadallah, S. J., & Gibson, G. (2008). A genome-wide gene expression signature of environmental geography in leukocytes of Moroccan Amazighs. PLoS Genetics, 4, e1000052.
Immordino-Yang, M. H., McColl, A., Damasio, H., & Damasio, A. (2009). Neural correlates of admiration and compassion. Proceedings of the National Academy of Sciences of the United States of America, 106, 8021–8026.
Irwin, M. R., & Cole, S. W. (2011). Reciprocal regulation of the neural and innate immune systems. Nature Reviews Immunology, 11, 625–632.
Irwin, M. R., Wang, M., Campomayor, C. O., Collado-Hidalgo, A., & Cole, S. (2006). Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation. Archives of Internal Medicine, 166, 1756–1762.
Karelina, K., Norman, G. J., Zhang, N., Morris, J. S., Peng, H., & DeVries, A. C. (2009). Social isolation alters neuroinflammatory response to stroke. Proceedings of the National Academy of Sciences of the United States of America, 106, 5895–5900.
Karssen, A. M., Her, S., Li, J. Z., Patel, P. D., Meng, F., Bunney, W. E. Jr., Jones, E. G., Watson, S. J., Akil, H., Myers, R. M., Schatzberg, A. F., & Lyons, D. M. (2007). Stress-induced changes in primate prefrontal profiles of gene expression. Molecular Psychiatry, 12, 1089–1102.
Kauffman, S. A. (1993). The origins of order: Self-organization and selection in evolution. New York: Oxford University Press.
Kawai, T., Morita, K., Masuda, K., Nishida, K., Shikishima, M., Ohta, M., Saito, T., & Rokutan, K. (2007). Gene expression signature in peripheral blood cells from medical students exposed to chronic psychological stress. Biological Psychology, 76, 147–155.
Keltner, D., Marsh, J., & Smith, J. A. (Eds.). (2010). The compassionate instinct: The science of human goodness. New York: W.W. Norton & Company.
Kemeny, M. E. (2009). Psychobiological responses to social threat: Evolution of a psychological model in psychoneuroimmunology. Brain, Behavior, and Immunity, 23, 1–9.
Kross, E., Berman, M. G., Mischel, W., Smith, E. E., & Wager, T. D. (2011). Social rejection shares somatosensory representations with physical pain. Proceedings of the National Academy of Sciences of the United States of America, 108, 6270–6275.
Lieberman, M. D. (2010). Social cognitive neuroscience. In S. T. Fiske, D. T. Gilbert, & G. Lindzey (Eds.), Handbook of Social Psychology, Fifth Edition (pp. 143–193). New York: McGraw-Hill.
Lieberman, M. D., & Eisenberger, N. I. (2009). Pains and pleasures of social life. Science, 323, 890–891.
Lutgendorf, S. K., De Geest, K., Bender, D., Ahmed, A., Goodheart, M. J., Dahmoush, L., Zimmerman, M. B., Penedo, F. J., Lucci, J. A., Ganjei-Azar, P., Thaker, P. H., Mendez, L., Lubaroff, D. M., Slavich, G. M., Cole, S. W., & Sood, A. K. (2012). Social influences on clinical outcome of ovarian cancer patients. Journal of Clinical Oncology, 30, 2885–2890.
Lutgendorf, S. K., DeGeest, K., Sung, C. Y., Arevalo, J. M., Penedo, F., Lucci J. A. III., Goodheart, M., Lubaroff, D., Farley, D. M., Sood, A. K., & Cole SW. (2009). Depression, social support, and beta-adrenergic transcription control in human ovarian cancer. Brain, Behavior, and Immunity, 23, 176–183.
Maranville, J. C., Luca, F., Richards, A. L., Wen, X., Witonsky, D. B., Baxter, S., Stephens, M., & Di Rienzo, A. (2011). Interactions between glucocorticoid treatment and cis-regulatory polymorphisms contribute to cellular response phenotypes. PLoS Genetics, 7, e1002162.
McEwen, B. S. (2007). Physiology and neurobiology of stress and adaptation: Central role of the brain. Physiological Reviews, 87, 873–904.
McEwen, B. S., & Gianaros, P. J. (2010). Central role of the brain in stress and adaptation: Links to socioeconomic status, health, and disease. Annals of the New York Academy of Sciences, 1186, 190–222.
McGowan, P. O., Sasaki, A., D’Alessio, A. C., Dymov, S., Labonté, B., Szyf, M., Turecki, G., & Meaney, M. J. (2009). Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nature Neuroscience, 12, 342–348.
Meaney, M. J., & Ferguson-Smith, A. C. (2010). Epigenetic regulation of the neural transcriptome: The meaning of the marks. Nature Neuroscience, 13, 1313–1318.
Medzhitov, R. (2008). Origin and physiological roles of inflammation. Nature, 454, 428–435.
Miller, G. E., & Chen, E. (2010). Harsh family climate in early life presages the emergence of a proinflammatory phenotype in adolescence. Psychological Science, 21, 848–856.
Miller, G., Chen, E., & Cole, S. W. (2009). Health psychology: Developing biologically plausible models linking the social world and physical health. Annual Review of Psychology, 60, 501–524.
Miller, G. E., Chen, E., Fok, A. K., Walker, H. A., Lim, A., Nicholls, E. F., Cole, S.W., & Kobor, M.S. (2009). Low early-life social class leaves a biological residue manifested by decreased glucocorticoid and increased proinflammatory signaling. Proceedings of the National Academy of Sciences of the United States of America, 106, 14716–14721.
Miller, G. E., Chen, E., & Parker, K. J. (2011). Psychological stress in childhood and susceptibility to the chronic diseases of aging: Moving towards a model of behavioral and biological mechanisms. Psychological Bulletin, 137, 959–997.
Miller, G. E., Chen, E., Sze, J., Marin, T., Arevalo, J. M., Doll, R., Ma, R., & Cole, S. W. (2008). A functional genomic fingerprint of chronic stress in humans: Blunted glucocorticoid and increased NF-κB signaling. Biological Psychiatry, 64, 266–272.
Miller, G. E., Rohleder, N., & Cole, S. W. (2009). Chronic interpersonal stress predicts activation of pro- and anti-inflammatory signaling pathways 6 months later. Psychosomatic Medicine, 71, 57–62.
Monroe, S. M., & Slavich, G. M. (2007). Psychological stressors, overview. In G. Fink (Ed.), Encyclopedia of Stress, Second Edition, Vol. 3 (pp. 278–284). Oxford: Academic Press.
Monroe, S. M., Slavich, G. M., & Georgiades, K. (2009). The social environment and life stress in depression. In I. H. Gotlib, & C. L. Hammen (Eds.), Handbook of Depression, Second Edition (pp. 340–360). New York: Guilford Press.
Murphy, M. L. M., Slavich, G. M., Rohleder, N., & Miller, G. E. (2013). Targeted rejection triggers differential pro- and anti-inflammatory gene expression in adolescents as a function of social status. Clinical Psychological Science, 1, 30–40.
Muscatell, K. A., & Eisenberger, N. I. (2012). A social neuroscience perspective on stress and health. Social and Personality Psychology Compass, 6, 890–904.
Muscatell, K. A., Morelli, S. A., Falk, E. B., Way, B. M., Pfeifer, J. H., Galinsky, A. D., Lieberman, M. D., Dapretto, M., & Eisenberger, N. I. (2012). Social status modulates neural activity in the mentalizing network. Neuroimage, 60, 1771–1777.
Nater, U. M., Whistler, T., Lonergan, W., Mletzko, T., Vernon, S. D., & Heim, C. (2009). Impact of acute psychosocial stress on peripheral blood gene expression pathways in healthy men. Biological Psychology, 82, 125–132.
Nolen-Hoeksema, S., Wisco, B. E., & Lyubomirsky, S. (2008). Rethinking rumination. Perspectives on Psychological Science, 3, 400–424.
O’Donovan, A., Hughes, B. M., Slavich, G. M., Lynch, L., Cronin, M. T., O’Farrelly, C., & Malone. K. M. (2010). Clinical anxiety, cortisol, and interleukin-6: Evidence for specificity in emotion-biology relationships. Brain, Behavior, and Immunity, 24, 1074–1077.
O’Donovan, A., Slavich, G. M., Epel, E. S., & Neylan, T. C. (2013). Exaggerated neurobiological sensitivity to threat as a mechanism linking anxiety with increased risk for diseases of aging. Neuroscience and Biobehavioral Reviews, 37, 96–108.
O’Donovan, A., Sun, B., Cole, S., Rempel, H., Lenoci, M., Pulliam, L., & Neylan, T. (2011). Transcriptional control of monocyte gene expression in post-traumatic stress disorder. Disease Markers, 30, 123–132.
Ornish, D., Magbanua, M. J., Weidner, G., Weinberg, V., Kemp, C., Green, C., Mattie, M. D., Marlin, R., Simko, J., Shinohara, K., Haqq, C. M., & Carroll, P. R. (2008). Changes in prostate gene expression in men undergoing an intensive nutrition and lifestyle intervention. Proceedings of the National Academy of Sciences of the United States of America, 105, 8369–8374.
Pace, T. W. W., Hu, F., & Miller, A. H. (2007). Cytokine-effects on glucocorticoid receptor function: Relevance to glucocorticoid resistance and the pathophysiology and treatment of major depression. Brain, Behavior, and Immunity, 21, 9–19.
Pace, T .W. W., Hu, F., & Miller, A. H. (2011). Activation of cAMP-protein kinase A abrogates STAT5-mediated inhibition of glucocorticoid receptor signaling by interferon-alpha. Brain, Behavior, and Immunity, 25, 1716–1724.
Pace, T. W. W., & Miller, A. H. (2009). Cytokines and glucocorticoid receptor signaling: Relevance to major depression. Annals of the New York Academy of Sciences, 1179, 86–105.
Panksepp, J. (1998). Affective neuroscience: The foundations of human and animal emotions. New York: Oxford University Press.
Raison, C. L., & Miller, A. H. (in press). The evolutionary significance of depression in Pathogen Host Defense (PATHOS-D). Molecular Psychiatry.
Rappaport, S. M., & Smith, M. T. (2010). Epidemiology. Environment and disease risks. Science, 330, 460–461.
Richerson, P. J., Boyd, R., & Henrich, J. (2010). Gene-culture coevolution in the age of genomics. Proceedings of the National Academy of Sciences of the United States of America, 107, 8985–8992.
Robinson, G. E. (2004). Genomics. Beyond nature and nurture. Science, 304, 397–399.
Robinson, G. E., Fernald, R. D., & Clayton, D. F. (2008). Genes and social behavior. Science, 322, 896–900.
Robinson, G. E., Grozinger, C. M., & Whitfield, C. W. (2005). Sociogenomics: Social life in molecular terms. Nature Reviews Genetics, 6, 257–270.
Sabeti, P. C., Schaffner, S. F., Fry, B., Lohmueller, J., Varilly, P., Shamovsky, O., Palma, A., Mikkelsen, T. S., Altshuler, D., & Lander, E. S. (2006). Positive natural selection in the human lineage. Science, 312, 1614–1620.
Shonkoff, J. P., Boyce, W. T., & McEwen, B. S. (2009). Neuroscience, molecular biology, and the childhood roots of health disparities: Building a new framework for health promotion and disease prevention. Journal of the American Medical Association, 301, 2252–2259.
Slavich, G. M., Monroe, S. M., & Gotlib, I. H. (2011). Early parental loss and depression history: Associations with recent life stress in major depressive disorder. Journal of Psychiatric Research, 45, 1146–1152.
Slavich, G. M., O’Donovan, A., Epel, E. S., & Kemeny, M. E. (2010). Black sheep get the blues: A psychobiological model of social rejection and depression. Neuroscience and Biobehavioral Reviews, 35, 39–45.
Slavich, G. M., Way, B. M., Eisenberger, N. I., & Taylor, S. E. (2010). Neural sensitivity to social rejection is associated with inflammatory responses to social stress. Proceedings of the National Academy of Sciences of the United States of America, 107, 14817–14822.
Slavich, G. M., Thornton, T., Torres, L. D., Monroe, S. M., & Gotlib, I. H. (2009). Targeted rejection predicts hastened onset of major depression. Journal of Social and Clinical Psychology, 28, 223–243.
Sloan, E. K., Capitanio, J. P., Tarara, R. P., & Cole, S. W. (2008). Social temperament and lymph node innervation. Brain, Behavior, and Immunity, 22, 717–726.
Sloan, E. K., Capitanio, J. P., Tarara, R. P., Mendoza, S. P., Mason, W. A., & Cole, S. W. (2007). Social stress enhances sympathetic innervation of primate lymph nodes: Mechanisms and implications for viral pathogenesis. Journal of Neuroscience, 27, 8857–8865.
Sloan, E. K., Priceman, S. J., Cox, B. F., Yu, S., Pimentel, M. A., Tangkanangnukul, V., Arevalo, J. M., Morizono, K., Karanikolas, B. D., Wu, L., Sood, A. K., & Cole, S. W. (2010). The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Research, 70, 7042–7052.
Stamou, P., & Kontoyiannis, D. L. (2010). Posttranscriptional regulation of TNF mRNA: A paradigm of signal-dependent mRNA utilization and its relevance to pathology. In G. Kollias, & P. P. Sfikakis (Eds.), TNF pathophysiology: Molecular and cellular mechanisms (pp. 61–79). Basel, Switzerland: Karger Publishers.
Sterling, P. (2004). Principles of allostasis: Optimal design, predictive regulation, pathophysiology, and rational therapeutics. In J. Schulkin (Ed.), Allostasis, homeostasis, and the costs of physiological adaptation (pp. 17–64). New York: Cambridge University Press.
Taylor, S. E. (2010). Mechanisms linking early life stress to adult health outcomes. Proceedings of the National Academy of Sciences of the United States of America, 107, 8507–8512.
Taylor, S. E., & Seeman, T. E. (1999). Psychosocial resources and the SES-health relationship. Annals of the New York Academy of Sciences, 896, 210–225.
Tsankova, N., Renthal, W., Kumar, A., & Nestler, E. J. (2007). Epigenetic regulation in psychiatric disorders. Nature Reviews Neuroscience, 8, 355–367.
Tung, J., Barreiro, L. B., Johnson, Z. P., Hansen, K. D., Michopoulos, V., Toufexis, D., Michelini, K., Wilson, M. E., & Gilad, Y. (2012). Social environment is associated with gene regulatory variation in the rhesus macaque immune system. Proceedings of the National Academy of Sciences of the United States of America, 109, 6490–6495.
Viger, R. S., Guittot, S. M., Anttonen, M., Wilson, D. B., Heikinheimo, M. (2008). Role of the GATA family of transcription factors in endocrine development, function, and disease. Molecular Endocrinology, 22, 781–798.
Virchow, R. (2006). Report on the typhus outbreak of upper silesia. Social Medicine, 1, 11–27. (Original work published 1848)
Way, B. M., & Taylor, S. E. (2010). Social influences on health: Is serotonin a critical mediator? Psychosomatic Medicine, 72, 107–112.
Weaver, I. C., Cervoni, N., Champagne, F. A., D’Alessio, A. C., Sharma, S., Seckl, J. R., Dymov, S., Szyf, M., & Meaney, M. J. (2004). Epigenetic programming by maternal behavior. Nature Neuroscience, 7, 847–854.
Weaver, I. C., Meaney, M. J., & Szyf, M. (2006). Maternal care effects on the hippocampal transcriptome and anxiety-mediated behaviors in the offspring that are reversible in adulthood. Proceedings of the National Academy of Sciences of the United States of America, 103, 3480–3485.
Webster, J. I., Tonelli, L., & Sternberg, E. M. (2002). Neuroendocrine regulation of immunity. Annual Review of Immunology, 20, 125–163.
Welle, S. (1999). Human protein metabolism. New York: Springer.
Williams, J. B., Pang, D., Delgado, B., Kocherginsky, M., Tretiakova, M., Krausz, T., Pan, D., He, J., McClintock, M. K., & Conzen, S. D. (2009). A model of gene-environment interaction reveals altered mammary gland gene expression and increased tumor growth following social isolation. Cancer Prevention Research, 2, 850–861.
Wohleb, E. S., Hanke, M. L., Corona, A. W., Powell, N. D., Stiner, L. M., Bailey, M. T., Nelson, R. J., Godbout, J. P., & Sheridan, J. F. (2011). β-Adrenergic receptor antagonism prevents anxiety-like behavior and microglial reactivity induced by repeated social defeat. Journal of Neuroscience, 31, 6277–6288.
Zieker, D., Fehrenbach, E., Dietzsch, J., Fliegner, J., Waidmann, M., Nieselt, K., Gebicke-Haerter, P., Spanagel, R., Simon, P., Niess, A. M., & Northoff, H. (2005). cDNA microarray analysis reveals novel candidate genes expressed in human peripheral blood following exhaustive exercise. Physiological Genomics, 23, 287–294.
